Get your patient on Chlorthalidone - Chlorthalidone tablet (Chlorthalidone)
Chlorthalidone - Chlorthalidone tablet prescribing information
INDICATIONS AND USAGE
Diuretics such as chlorthalidone are indicated in the management of hypertension either as the sole therapeutic agent or to enhance the effect of other antihypertensive drugs in the more severe forms of hypertension.
Chlorthalidone is indicated as adjunctive therapy in edema associated with congestive heart failure, hepatic cirrhosis, and corticosteroid and estrogen therapy.
Chlorthalidone has also been found useful in edema due to various forms of renal dysfunction, such as nephrotic syndrome, acute glomerulonephritis, and chronic renal failure.
DOSAGE AND ADMINISTRATION
Therapy should be initiated with the lowest possible dose, then titrated according to individual patient response. A single dose given in the morning with food is recommended; divided daily doses are unnecessary.
CONTRAINDICATIONS
Anuria.
Known hypersensitivity to chlorthalidone or other sulfonamide-derived drugs.
ADVERSE REACTIONS
The following adverse reactions have been observed, but there is not enough systematic collection of data to support an estimate of their frequency.
Gastrointestinal System Reactions: anorexia, gastric irritation, nausea, vomiting, cramping, diarrhea, constipation, jaundice (intrahepatic cholestatic jaundice), pancreatitis.
Central Nervous System Reactions: dizziness, vertigo, paresthesias, headache, xanthopsia.
Hematologic Reactions: leukopenia, agranulocytosis, thrombocytopenia, aplastic anemia.
Dermatologic-Hypersensitivity Reactions: purpura, photosensitivity, rash, urticaria, necrotizing angiitis (vasculitis, cutaneous vasculitis), Lyell’s syndrome (toxic epidermal necrolysis).
Cardiovascular Reactions: orthostatic hypotension may occur and may be aggravated by alcohol, barbiturates, or narcotics.
Other Adverse Reactions: hyperglycemia, glycosuria, hyperuricemia, muscle spasm, weakness, restlessness, impotence.
Whenever adverse reactions are moderate or severe, chlorthalidone dosage should be reduced or therapy withdrawn.
DESCRIPTION
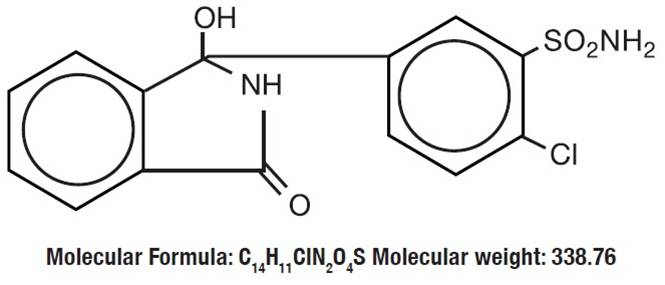
Chlorthalidone is an oral antihypertensive/diuretic. It is a monosulfamyl diuretic that differs chemically from thiazide diuretics in that a double-ring system is incorporated in its structure. It is 2-Chloro-5(1-hydroxy-3-oxo-1-isoindolinyl) benzenesulfonamide with the following structural formula:
Chlorthalidone, USP is practically insoluble in water, in ether, and in chloroform; soluble in methanol; slightly soluble in ethanol. Chlorthalidone tablets are available containing either 25 mg or 50 mg of chlorthalidone, USP and the following inactive ingredients: colloidal silicon dioxide, microcrystalline cellulose, pregelatinized starch (corn), sodium starch glycolate, stearic acid.
CLINICAL PHARMACOLOGY
Chlorthalidone is an oral diuretic with prolonged action (48–72 hours) and low toxicity. The major portion of the drug is excreted unchanged by the kidneys. The diuretic effect of the drug occurs in approximately 2.6 hours and continues for up to 72 hours. The mean half-life following a 50 to 200 mg dose is 40 hours. In the first order of absorption, the elimination half-life is 53 hours following a 50 mg dose, and 60 hours following a 100 mg dose. Approximately 75 percent of the drug is bound to plasma proteins, 58 percent of the drug being bound to albumin. This is caused by an increased affinity of the drug to erythrocyte carbonic anhydrase. Nonrenal routes of elimination have yet to be clarified. Data are not available regarding percentage of dose as unchanged drug and metabolites, concentration of the drug in body fluids, degree of uptake by a particular organ or in the fetus, or passage across the blood-brain barrier.
The drug produces copious diuresis with greatly increased excretion of sodium and chloride. At maximal therapeutic dosage, chlorthalidone is approximately equal in its diuretic effect to comparable maximal therapeutic doses of benzothiadiazine diuretics. The site of action appears to be the cortical diluting segment of the ascending limb of Henle’s loop of the nephron.
HOW SUPPLIED
Chlorthalidone Tablets, USP are available containing 25 mg or 50 mg of Chlorthalidone, USP.
The 25 mg tablets are white to off white, round shaped, flat faced, uncoated tablet with ‘ ET30 ’ debossed on one side and plain on other side. They are available as follows:
NDC 62135-505-30 Bottles of 30
NDC 62135-505-90 Bottles of 90
The 50 mg tablets are white to off white, round shaped, flat faced, uncoated tablet with break line and ‘ ET31 ’ debossed on one side and plain on other side. They are available as follows:
NDC 62135-506-30 Bottles of 30
NDC 62135-506-90 Bottles of 90
Store at 20° to 25°C (68° to 77°F). [See USP for Controlled Room Temperature.]
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.