Get your patient on Nystatin - Nystatin suspension (Nystatin)
Nystatin - Nystatin suspension prescribing information
INDICATIONS AND USAGE
Nystatin oral suspension is indicated for the treatment of infections of the oral cavity caused by Candida albicans .
DOSAGE AND ADMINISTRATION
Infants: 2 mL (200,000 units) four times daily (1 mL in each side of mouth).
Pediatric patients and adults: 4 to 6 mL (400,000 to 600,000 units) four times daily (one-half of dose in each side of mouth).
NOTE: Limited clinical studies in neonates, including premature and low-birth weight neonates, indicate that 1 mL (100,000 units) four times daily is effective.
Local treatment should be continued at least 48 hours after perioral symptoms have disappeared and/or cultures returned to normal. It is recommended that the drug be retained in the mouth as long as possible before swallowing.
CONTRAINDICATIONS
Nystatin is contraindicated in patients with a history of hypersensitivity to nystatin or any of the suspension components.
ADVERSE REACTIONS
Gastrointestinal symptoms including diarrhea, gastrointestinal distress, nausea, vomiting and burning of the mouth have been reported. Hypersensitivity reactions including rash, pruritus, and anaphylactoid reaction have also been reported.
DESCRIPTION
Nystatin, USP is obtained from Streptomyces noursei . It is known to be a mixture, but the composition has not been completely elucidated. Nystatin A is closely related to amphotericin B. Each is a macro-cyclic lactone containing a ketal ring, an all-trans polyene system, and a mycosamine (3-amino-3-deoxyrhamose) moiety.
Its structural formula is:
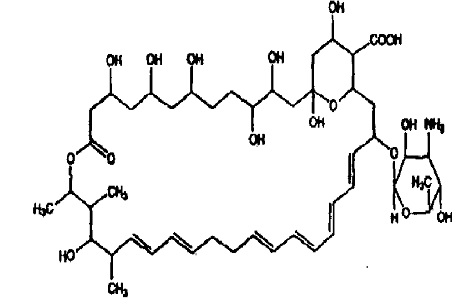
C 47 H 75 NO 17 M.W .926 .13
Nystatin Oral Suspension, USP, is a cherry-flavored, ready-to-use suspension containing 100,000 units of Nystatin, USP per mL. Nystatin, USP contains the following inactive ingredients: artificial (wild) cherry flavor, D&C Yellow 10, edetate calcium disodium, hydrochloric acid, methylparaben, polysorbate 80, propylparaben, purified bentonite, purified water, sodium hydroxide and sucrose.
CLINICAL PHARMACOLOGY
Nystatin acts by binding to sterols in the cell membrane of the fungus with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin is absorbed very sparingly following oral administration, with no detectable blood levels when given in the recommended doses.
HOW SUPPLIED
Nystatin Oral Suspension, USP is a bright yellow color suspension with cherry flavor containing 100,000 units of nystatin per mL, supplied as follows:
NDC 62135-813-46 – bottle of 60 mL with dropper
NDC 62135-813-47 – bottle of 473 mL
Store at controlled room temperature between 20°C to 25°C (68°F to 77°F).
DO NOT FREEZE
Manufactured for: Chartwell RX, LLC. Congers, NY 10920
L71884
Rev. 03/2026