Get your patient on Penicillamine - Penicillamine capsule (Penicillamine)
Penicillamine - Penicillamine capsule prescribing information
INDICATIONS
Penicillamine capsules are indicated in the treatment of Wilson's disease, cystinuria, and in patients with severe, active rheumatoid arthritis who have failed to respond to an adequate trial of conventional therapy. Available evidence suggests that penicillamine capsules are not of value in ankylosing spondylitis.
Wilson’s Disease
Wilson's disease (hepatolenticular degeneration) occurs in individuals who have inherited an autosomal-recessive defect that leads to an accumulation of copper far in excess of metabolic requirements. The excess copper is deposited in several organs and tissues, and eventually produces pathological effects primarily in the liver, where damage progresses to postnecrotic cirrhosis, and in the brain, where degeneration is widespread. Copper is also deposited as characteristic, asymptomatic, golden-brown Kayser-Fleischer rings in the corneas of all patients with cerebral symptomatology and some patients who are either asymptomatic or manifest only hepatic symptomatology.
Two types of patients require treatment for Wilson's disease: (1) the symptomatic, and (2) the asymptomatic in whom it can be assumed the disease will develop in the future if the patient is not treated.
The diagnosis, if suspected on the basis of family or individual history or physical examination, can be confirmed if the plasma copper-protein ceruloplasmin•• is less than 20 mg/dL and either a quantitative determination in a liver biopsy specimen shows an abnormally high concentration of copper (greater than 250 mcg/g dry weight) or Kayser-Fleischer rings are present.
Treatment has two objectives:
(1) to minimize dietary intake of copper;
(2) to promote excretion and complex formation (i.e., detoxification) of excess tissue copper.
The first objective is attained by a daily diet that contains no more than 1 or 2 mg of copper. Such a diet should exclude, most importantly, chocolate, nuts, shellfish, mushrooms, liver, molasses, broccoli, and cereals and dietary supplements enriched with copper, and be composed to as great an extent as possible of foods with a low copper content. Distilled or demineralized water should be used if the patient's drinking water contains more than 0.1 mg/L of copper.
For the second objective, a copper chelating agent is used.
In symptomatic patients, this treatment usually produces marked neurologic improvement, fading of Kayser-Fleischer rings, and gradual amelioration of hepatic dysfunction and psychic disturbances.
Clinical experience to date suggests that life is prolonged with the above regimen.
Noticeable improvement may not occur for 1 to 3 months. Occasionally, neurologic symptoms become worse during initiation of therapy with penicillamine capsules. Despite this, the drug should not be withdrawn. Temporary interruption carries an increased risk of developing a sensitivity reaction upon resumption of therapy, although it may result in clinical improvement of neurological symptoms (see WARNINGS ). If the neurological symptoms and signs continue to worsen for a month after the initiation of penicillamine capsules therapy, several short courses of treatment with 2,3-dimercaprol (BAL) while continuing penicillamine capsules may be considered.
Treatment of asymptomatic patients has been carried out for over 30 years. Symptoms and signs of the disease appear to be prevented indefinitely if daily treatment with penicillamine capsules are continued.
Cystinuria
Cystinuria is characterized by excessive urinary excretion of the dibasic amino acids, arginine, lysine, ornithine, and cystine, and the mixed disulfide of cysteine and homocysteine. The metabolic defect that leads to cystinuria is inherited as an autosomal-recessive trait. Metabolism of the affected amino acids is influenced by at least two abnormal factors: (1) defective gastrointestinal absorption and (2) renal tubular dysfunction.
Arginine, lysine, ornithine, and cysteine are soluble substances, readily excreted. There is no apparent pathology connected with their excretion in excessive quantities.
Cystine, however, is so slightly soluble at the usual range of urinary pH that it is not excreted readily, and so crystallizes and forms stones in the urinary tract. Stone formation is the only known pathology in cystinuria.
Normal daily output of cystine is 40 to 80 mg. In cystinuria, output is greatly increased and may exceed 1 g/day. At 500 to 600 mg/day, stone formation is almost certain. When it is more than 300 mg/day, treatment is indicated.
Conventional treatment is directed at keeping urinary cystine diluted enough to prevent stone formation, keeping the urine alkaline enough to dissolve as much cystine as possible, and minimizing cystine production by a diet low in methionine (the major dietary precursor of cystine). Patients must drink enough fluid to keep urine-specific gravity below 1.010, take enough alkali to keep urinary pH at 7.5 to 8, and maintain a diet low in methionine. This diet is not recommended in growing children and probably is contraindicated in pregnancy because of its low protein content (see PRECAUTIONS ).
When these measures are inadequate to control recurrent stone formation, penicillamine capsules may be used as additional therapy, and when patients refuse to adhere to conventional treatment, penicillamine capsules may be a useful substitute. It is capable of keeping cystine excretion to near normal values, thereby hindering stone formation and the serious consequences of pyelonephritis and impaired renal function that develop in some patients. Bartter and colleagues depict the process by which penicillamine interacts with cystine to form penicillamine-cysteine mixed disulfide as:
CSSC + PS'  CS' + CSSP
CS' + CSSP
PSSP + CS'  PS' + CSSP
PS' + CSSP
CSSC + PSSP'  2CSSP
2CSSP
CSSC = cystine
CS' = deprotonated cysteine
PSSP = penicillamine disulfide
PS' = deprotonated penicillamine sulfhydryl
CSSP = penicillamine-cysteine mixed disulfide
In this process, it is assumed that the deprotonated form of penicillamine, PS', is the active factor in bringing about the disulfide interchange.
Rheumatoid Arthritis
Because penicillamine capsules can cause severe adverse reactions, its use in rheumatoid arthritis should be restricted to patients who have severe, active disease and who have failed to respond to an adequate trial of conventional therapy. Even then, benefit-to-risk ratio should be carefully considered. Other measures, such as rest, physiotherapy, salicylates, and corticosteroids, should be used, when indicated, in conjunction with penicillamine capsules (see PRECAUTIONS ).
DOSAGE AND ADMINISTRATION
In all patients receiving penicillamine, it is important that penicillamine capsules be given on an empty stomach, at least one hour before meals or two hours after meals, and at least one hour apart from any other drug, food, or milk. Because penicillamine increases the requirement for pyridoxine, patients may require a daily supplement of pyridoxine (see PRECAUTIONS ).
Wilson’s Disease
Optimal dosage can be determined by measurement of urinary copper excretion and the determination of free copper in the serum. The urine must be collected in copper-free glassware, and should be quantitatively analyzed for copper before and soon after initiation of therapy with penicillamine capsules.
Determination of 24-hour urinary copper excretion is of greatest value in the first week of therapy with penicillamine. In the absence of any drug reaction, a dose between 0.75 and 1.5 g that results in an initial 24-hour cupriuresis of over 2 mg should be continued for about 3 months, by which time the most reliable method of monitoring maintenance treatment is the determination of free copper in the serum. This equals the difference between quantitatively determined total copper and ceruloplasmin copper. Adequately treated patients will usually have less than 10 mcg free copper/dL of serum. It is seldom necessary to exceed a dosage of 2 g/day. If the patient is intolerant to therapy with penicillamine capsules, alternative treatment is trientine hydrochloride.
In patients who cannot tolerate as much as 1 g/day initially, initiating dosage with 250 mg/day and increasing gradually to the requisite amount, gives closer control of the effects of the drug and may help to reduce the incidence of adverse reactions.
Cystinuria
It is recommended that penicillamine capsules be used along with conventional therapy. By reducing urinary cystine, it decreases crystalluria and stone formation. In some instances, it has been reported to decrease the size of, and even to dissolve, stones already formed.
The usual dosage of penicillamine capsules in the treatment of cystinuria is 2 g/day for adults, with a range of 1 to 4 g/day. For pediatric patients, dosage can be based on 30 mg/kg/day. The total daily amount should be divided into four doses. If four equal doses are not feasible, give the larger portion at bedtime. If adverse reactions necessitate a reduction in dosage, it is important to retain the bedtime dose.
Initiating dosage with 250 mg/day, and increasing gradually to the requisite amount, gives closer control of the effects of the drug and may help to reduce the incidence of adverse reactions.
In addition to taking penicillamine capsules, patients should drink copiously. It is especially important to drink about a pint of fluid at bedtime and another pint once during the night when urine is more concentrated and more acid than during the day. The greater the fluid intake, the lower the required dosage of penicillamine capsules.
Dosage must be individualized to an amount that limits cystine excretion to 100-200 mg/day in those with no history of stones, and below 100 mg/day in those who have had stone formation and/or pain. Thus, in determining dosage, the inherent tubular defect, the patient's size, age, and rate of growth, and his diet and water intake all must be taken into consideration.
The standard nitroprusside cyanide test has been reported useful as a qualitative measure of the effective dose:† Add 2 mL of freshly prepared 5% sodium cyanide to 5 mL of a 24-hour aliquot of protein-free urine and let stand 10 minutes. Add five drops of freshly prepared 5% sodium nitroprusside and mix. Cystine will turn the mixture magenta. If the result is negative, it can be assumed that cystine excretion is less than 100 mg/g creatinine.
Although penicillamine is rarely excreted unchanged, it also will turn the mixture magenta. If there is any question as to which substance is causing the reaction, a ferric chloride test can be done to eliminate doubt: Add 3% ferric chloride dropwise to the urine. Penicillamine will turn the urine an immediate and quickly fading blue. Cystine will not produce any change in appearance.
Rheumatoid Arthritis
The principal rule of treatment with penicillamine capsules in rheumatoid arthritis is patience. The onset of therapeutic response is typically delayed. Two or three months may be required before the first evidence of a clinical response is noted (see CLINICAL PHARMACOLOGY ).
When treatment with penicillamine capsules has been interrupted because of adverse reactions or other reasons, the drug should be reintroduced cautiously by starting with a lower dosage and increasing slowly.
Initial Therapy
The currently recommended dosage regimen in rheumatoid arthritis begins with a single daily dose of 125 mg or 250 mg, which is thereafter increased at 1- to 3-month intervals, by 125 mg or 250 mg/day, as patient response and tolerance indicate. If a satisfactory remission of symptoms is achieved, the dose associated with the remission should be continued (see DOSAGE AND ADMINISTRATION, Rheumatoid Arthritis, Maintenance Therapy ). If there is no improvement and there are no signs of potentially serious toxicity after 2 to 3 months of treatment with doses of 500-750 mg/day, increases of 250 mg/day at 2- to 3-month intervals may be continued until a satisfactory remission occurs (see DOSAGE AND ADMINISTRATION, Rheumatoid Arthritis, Maintenance Therapy ) or signs of toxicity develop (see WARNINGS and PRECAUTIONS ). If there is no discernible improvement after three to four months of treatment with 1000 to 1500 mg of penicillamine/day, it may be assumed the patient will not respond and penicillamine capsules should be discontinued.
Maintenance Therapy
The maintenance dosage of penicillamine capsules must be individualized, and may require adjustment during the course of treatment. Many patients respond satisfactorily to a dosage within the 500 to 750 mg/day range. Some need less.
Changes in maintenance dosage levels may not be reflected clinically or in the erythrocyte sedimentation rate for 2 to 3 months after each dosage adjustment.
Some patients will subsequently require an increase in the maintenance dosage to achieve maximal disease suppression. In those patients who do respond, but who evidence incomplete suppression of their disease after the first 6 to 9 months of treatment, the daily dosage of penicillamine capsules may be increased by 125 mg or 250 mg/day at 3-month intervals. It is unusual in current practice to employ a dosage in excess of 1 g/day, but up to 1.5 g/day has sometimes been required.
Management of Exacerbations
During the course of treatment some patients may experience an exacerbation of disease activity following an initial good response. These may be self-limited and can subside within 12 weeks. They are usually controlled by the addition of non-steroidal anti-inflammatory drugs, and only if the patient has demonstrated a true "escape" phenomenon (as evidenced by failure of the flare to subside within this time period) should an increase in the maintenance dose ordinarily be considered.
In the rheumatoid patient, migratory polyarthralgia due to penicillamine is extremely difficult to differentiate from an exacerbation of rheumatoid arthritis. Discontinuance or a substantial reduction in dosage of penicillamine capsules for up to several weeks will usually determine which of these processes is responsible for the arthralgia.
Duration of Therapy
The optimum duration of therapy with penicillamine capsules in rheumatoid arthritis has not been determined. If the patient has been in remission for 6 months or more, a gradual, stepwise dosage reduction in decrements of 125 mg or 250 mg/day at approximately 3-month intervals may be attempted.
Concomitant Drug Therapy
Penicillamine Capsules should not be used in patients who are receiving gold therapy, antimalarial or cytotoxic drugs, oxyphenbutazone, or phenylbutazone (see PRECAUTIONS ). Other measures, such as salicylates, other non-steroidal anti-inflammatory drugs, or systemic corticosteroids, may be continued when penicillamine is initiated. After improvement commences, analgesic and anti-inflammatory drugs may be slowly discontinued as symptoms permit. Steroid withdrawal must be done gradually, and many months of treatment with penicillamine capsules may be required before steroids can be completely eliminated.
Dosage Frequency
Based on clinical experience, dosages up to 500 mg/day can be given as a single daily dose. Dosages in excess of 500 mg/day should be administered in divided doses.
CONTRAINDICATIONS
Except for the treatment of Wilson's disease or certain patients with cystinuria, use of penicillamine during pregnancy is contraindicated (see WARNINGS ).
Although breast milk studies have not been reported in animals or humans, mothers on therapy with penicillamine should not nurse their infants.
Patients with a history of penicillamine-related aplastic anemia or agranulocytosis should not be restarted on penicillamine (see WARNINGS and ADVERSE REACTIONS ).
Because of its potential for causing renal damage, penicillamine should not be administered to rheumatoid arthritis patients with a history or other evidence of renal insufficiency.
ADVERSE REACTIONS
Penicillamine is a drug with a high incidence of untoward reactions, some of which are potentially fatal. Therefore, it is mandatory that patients receiving penicillamine therapy remain under close medical supervision throughout the period of drug administration (see WARNINGS and PRECAUTIONS ).
Reported incidences (%) for the most commonly occurring adverse reactions in rheumatoid arthritis patients are noted, based on 17 representative clinical trials reported in the literature (1270 patients).
Allergic
Generalized pruritus, early and late rashes (5%), pemphigus (see WARNINGS ), and drug eruptions, which may be accompanied by fever, arthralgia, or lymphadenopathy have occurred (see WARNINGS and PRECAUTIONS ). Some patients may show a lupus erythematosus-like syndrome similar to drug-induced lupus produced by other pharmacological agents (see PRECAUTIONS ).
Urticaria and exfoliative dermatitis have occurred.
Thyroiditis has been reported; hypoglycemia in association with anti-insulin antibodies has been reported. These reactions are extremely rare.
Some patients may develop a migratory polyarthralgia, often with objective synovitis (see DOSAGE AND ADMINISTRATION ).
Gastrointestinal
Anorexia, epigastric pain, nausea, vomiting, or occasional diarrhea may occur (17%).
Isolated cases of reactivated peptic ulcer have occurred, as have hepatic dysfunction including hepatic failure and pancreatitis. Intrahepatic cholestasis and toxic hepatitis have been reported rarely. There have been a few reports of increased serum alkaline phosphatase, lactic dehydrogenase, and positive cephalin flocculation and thymol turbidity tests.
Some patients may report a blunting, diminution, or total loss of taste perception (12%) or may develop oral ulcerations. Although rare, cheilosis, glossitis, and gingivostomatitis have been reported (see PRECAUTIONS ).
Gastrointestinal side effects are usually reversible following cessation of therapy.
Hematological
Penicillamine can cause bone marrow depression (see WARNINGS ). Leukopenia (2%) and thrombocytopenia (4%) have occurred. Fatalities have been reported as a result of thrombocytopenia, agranulocytosis, aplastic anemia, and sideroblastic anemia.
Thrombotic thrombocytopenic purpura, hemolytic anemia, red cell aplasia, monocytosis, leukocytosis, eosinophilia, and thrombocytosis have also been reported.
Renal
Patients on penicillamine therapy may develop proteinuria (6%) and/or hematuria which, in some, may progress to the development of the nephrotic syndrome as a result of an immune complex membranous glomerulopathy (see WARNINGS ). Renal failure has been reported.
Central Nervous System
Tinnitus, optic neuritis, and peripheral sensory and motor neuropathies (including polyradiculoneuropathy, i.e., Guillain-Barr¡SR syndrome) have been reported. Muscular weakness may or may not occur with the peripheral neuropathies. Visual and psychic disturbances; mental disorders; and agitation and anxiety have been reported.
Neuromuscular
Myasthenia gravis (see WARNINGS ); dystonia.
Other
Adverse reactions that have been reported rarely include thrombophlebitis; hyperpyrexia (see PRECAUTIONS ); falling hair or alopecia; lichen planus; polymyositis; dermatomyositis; mammary hyperplasia; elastosis perforans serpiginosa; toxic epidermal necrolysis; anetoderma (cutaneous macular atrophy); and Goodpasture's syndrome, a severe and ultimately fatal glomerulonephritis associated with intra-alveolar hemorrhage (see WARNINGS ). Vasculitis, including fatal renal vasculitis, has also been reported. Allergic alveolitis, obliterative bronchiolitis, interstitial pneumonitis, and pulmonary fibrosis have been reported in patients with severe rheumatoid arthritis, some of whom were receiving penicillamine. Bronchial asthma also has been reported.
Increased skin friability; excessive wrinkling of skin; and development of small white papules at venipuncture and surgical sites have been reported (see PRECAUTIONS ); yellow nail syndrome.
The chelating action of the drug may cause increased excretion of other heavy metals such as zinc, mercury, and lead.
There have been reports associating penicillamine with leukemia. However, circumstances involved in these reports are such that a cause and effect relationship to the drug has not been established.
To report SUSPECTED ADVERSE REACTIONS, contact Navinta LLC at +1-609-883-1135 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Penicillamine is a chelating agent used in the treatment of Wilson's disease. It is also used to reduce cystine excretion in cystinuria and to treat patients with severe, active rheumatoid arthritis unresponsive to conventional therapy (see INDICATIONS ). It is 3-mercapto-D-valine. It is a white, or practically white, crystalline powder, freely soluble in water, slightly soluble alcohol, insoluble in chloroform and in ether. Although its configuration is D, it is levorotatory as usually measured:
[α] 25°= -62.5° ± 2° (c = 1, 1N NaOH),
D
calculated on a dried basis.
The empirical formula is C 5 H 11 NO 2 S, giving it a molecular weight of 149.21. The structural formula is:
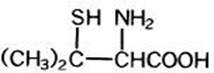
It reacts readily with formaldehyde or acetone to form a thiazolidine-carboxylic acid. Penicillamine Capsules, USP for oral administration contain 250 mg of penicillamine. Each capsule contains the following inactive ingredients: lactose monohydrate, magnesium stearate, D&C yellow no. 10, gelatin and titanium dioxide. The imprinting ink contains shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, black iron oxide, potassium hydroxide and purified water.
CLINICAL PHARMACOLOGY
Penicillamine is a chelating agent recommended for the removal of excess copper in patients with Wilson's disease. From in vitro studies which indicate that one atom of copper combines with two molecules of penicillamine, it would appear that 1 g of penicillamine should be followed by the excretion of about 200 mg of copper; however, the actual amount excreted is about 1% of this.
Penicillamine also reduces excess cystine excretion in cystinuria. This is done, at least in part, by disulfide interchange between penicillamine and cystine, resulting in formation of penicillamine-cysteine disulfide, a substance that is much more soluble than cystine and is excreted readily.
Penicillamine interferes with the formation of cross-links between tropocollagen molecules and cleaves them when newly formed.
The mechanism of action of penicillamine in rheumatoid arthritis is unknown although it appears to suppress disease activity. Unlike cytotoxic immunosuppressants, penicillamine markedly lowers IgM rheumatoid factor but produces no significant depression in absolute levels of serum immunoglobulins. Also unlike cytotoxic immunosuppressants which act on both, penicillamine in vitro depresses T-cell activity but not B-cell activity.
In vitro , penicillamine dissociates macroglobulins (rheumatoid factor) although the relationship of the activity to its effect in rheumatoid arthritis is not known.
In rheumatoid arthritis, the onset of therapeutic response to penicillamine may not be seen for 2 or 3 months. In those patients who respond, however, the first evidence of suppression of symptoms such as pain, tenderness, and swelling is generally apparent within 3 months. The optimum duration of therapy has not been determined. If remissions occur, they may last from months to years, but usually require continued treatment (see DOSAGE AND ADMINISTRATION ).
In all patients receiving penicillamine, it is important that penicillamine capsules be given on an empty stomach, at least 1 hour before meals or 2 hours after meals, and at least 1 hour apart from any other drug, food, milk, antacid, zinc, or iron-containing preparation. This permits maximum absorption and reduces the likelihood of inactivation by metal binding in the gastrointestinal tract.
Pharmacokinetics
Penicillamine is absorbed rapidly but incompletely (40-70%) from the gastrointestinal tract, with wide inter-individual variations. Food, antacids, and iron reduce absorption of the drug. The peak plasma concentration of penicillamine occurs 1 to 3 hours after ingestion; it is approximately 1 to 2 mg/L after an oral dose of 250 mg. The drug appears in the plasma as free penicillamine, penicillamine disulfide, and penicillamine-cysteine disulfide. When prolonged treatment is stopped, there is a slow elimination phase lasting 4 to 6 days.
More than 80% of plasma penicillamine is bound to proteins, especially albumin and ceruloplasmin. The drug also binds to erythrocytes and macrophages. A small fraction of the dose is metabolized in the liver to S-methyl-D-penicillamine. Excretion is mainly renal, mainly as disulfides.
HOW SUPPLIED
Penicillamine Capsules USP, 250 mg are opaque ivory colored hard gelatin capsules containing white to off white powder and are imprinted with "114" and "NAV" in black ink. They are supplied as follows:
NDC 68475-201-01 in bottles of 100.
Storage
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Dispense in a tightly closed container. Keep container tightly closed.
Manufactured for:
Navinta LLC
1499 lower Ferry Road, Ewing,
NJ 08618
Made in India
Rev. 10/2021
31141021 R0
•• For quantitative test for serum ceruloplasmin see: Morell, A.G.; Windsor, J.; Sternlieb, I. ; Scheinberg, I.H.: Measurement of the concentration of ceruloplasmin in serum by determination of its oxidase activity, in "Laboratory Diagnosis of Liver Disease", F.W. Sunderman; F.W. Sunderman, Jr. (eds.), St. Louis, Warren H. Green, Inc., 1968, pp. 193-195.
••• Scheinberg, I.H.; Sternlieb, I.: N. Engl. J. Med. 293: 1300-1302, Dec. 18, 1975.
† Lotz, M.; Potts, J.T. and Bartter, F.C.: Brit. Med. J. 2: 521, Aug. 28, 1965 (in Medical Memoranda).